Closed & Open Systems | Definition, Differences & Thermodynamics - Video & Lesson Transcript | Study.com

Define a system and simplify the open-system energy balance for each of the following cases - YouTube

With the help of a diagram, explain the concept of an organization as an open system. | Homework.Study.com
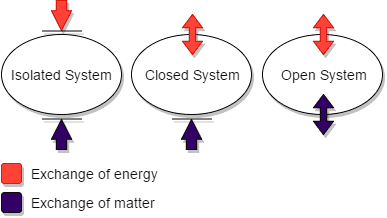
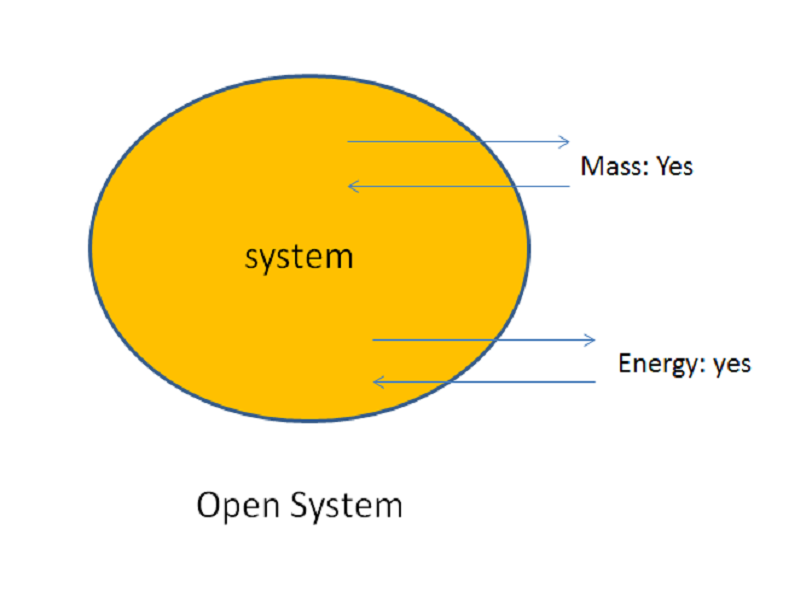
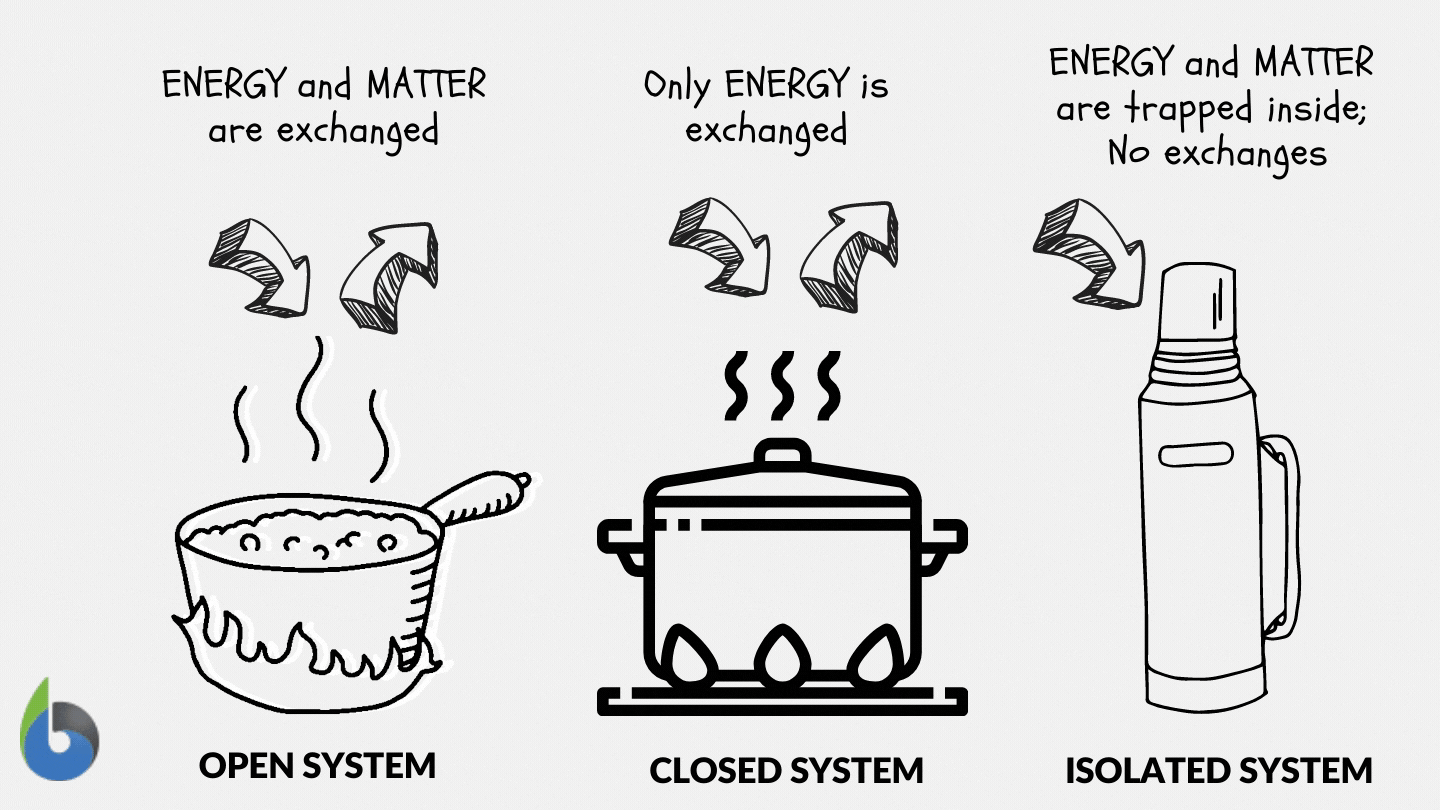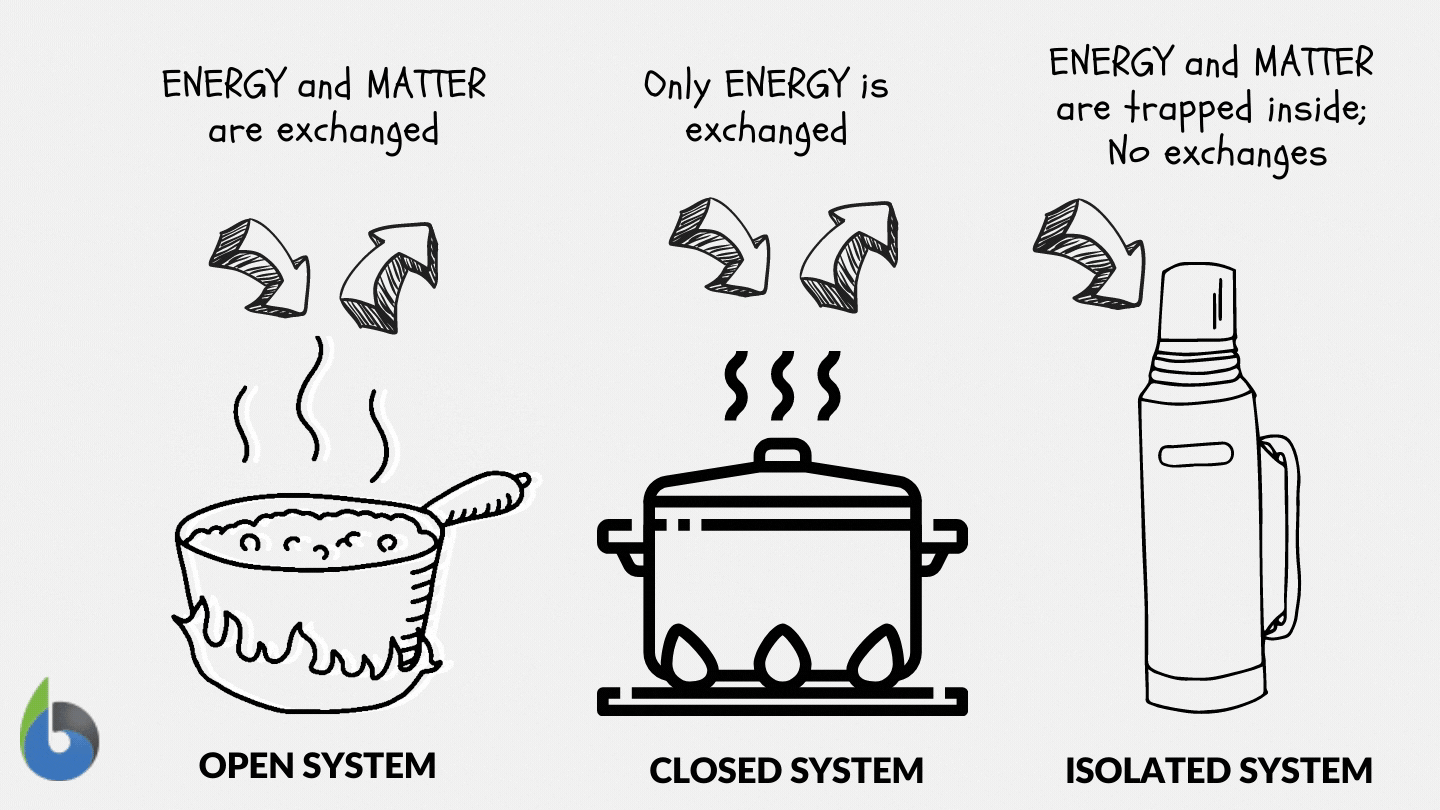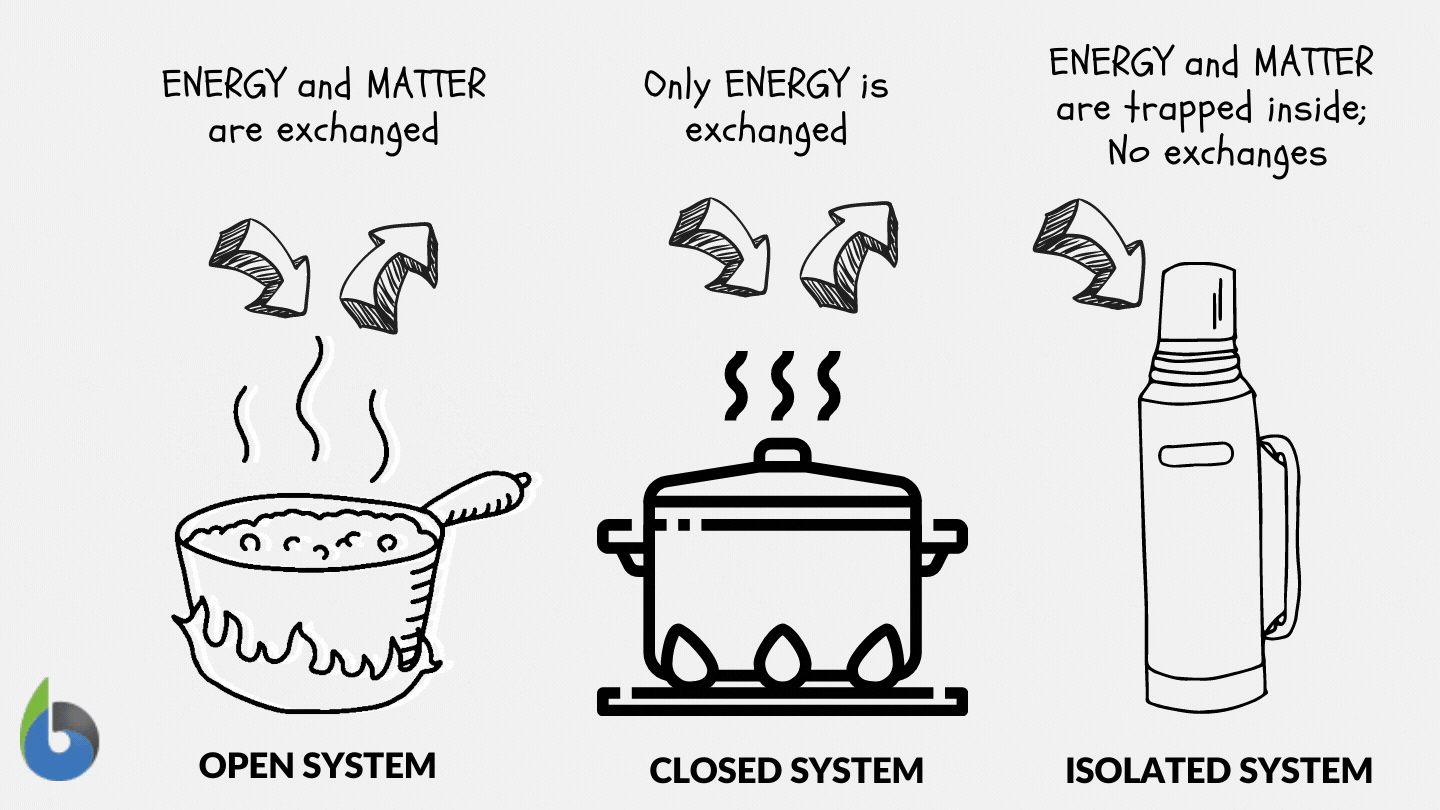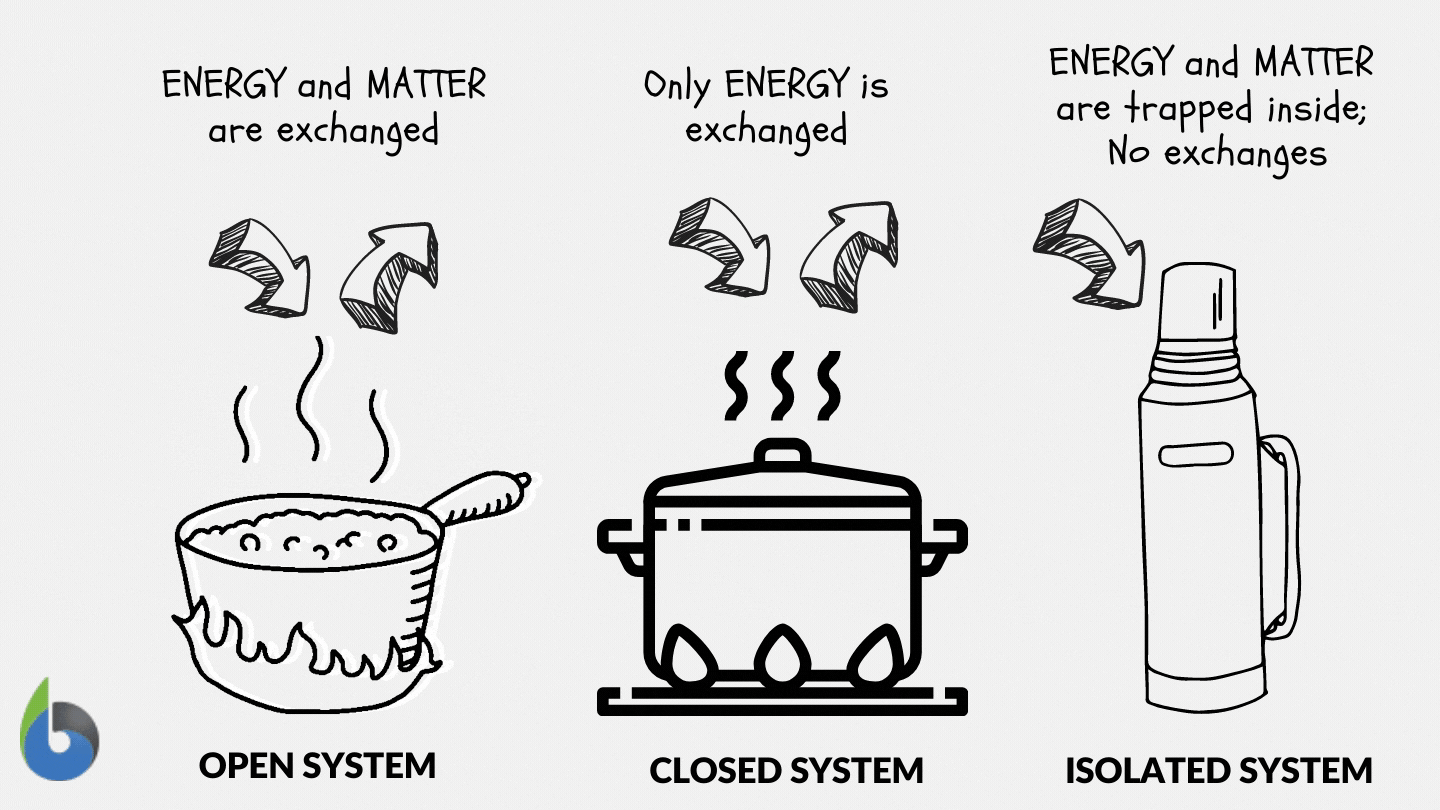
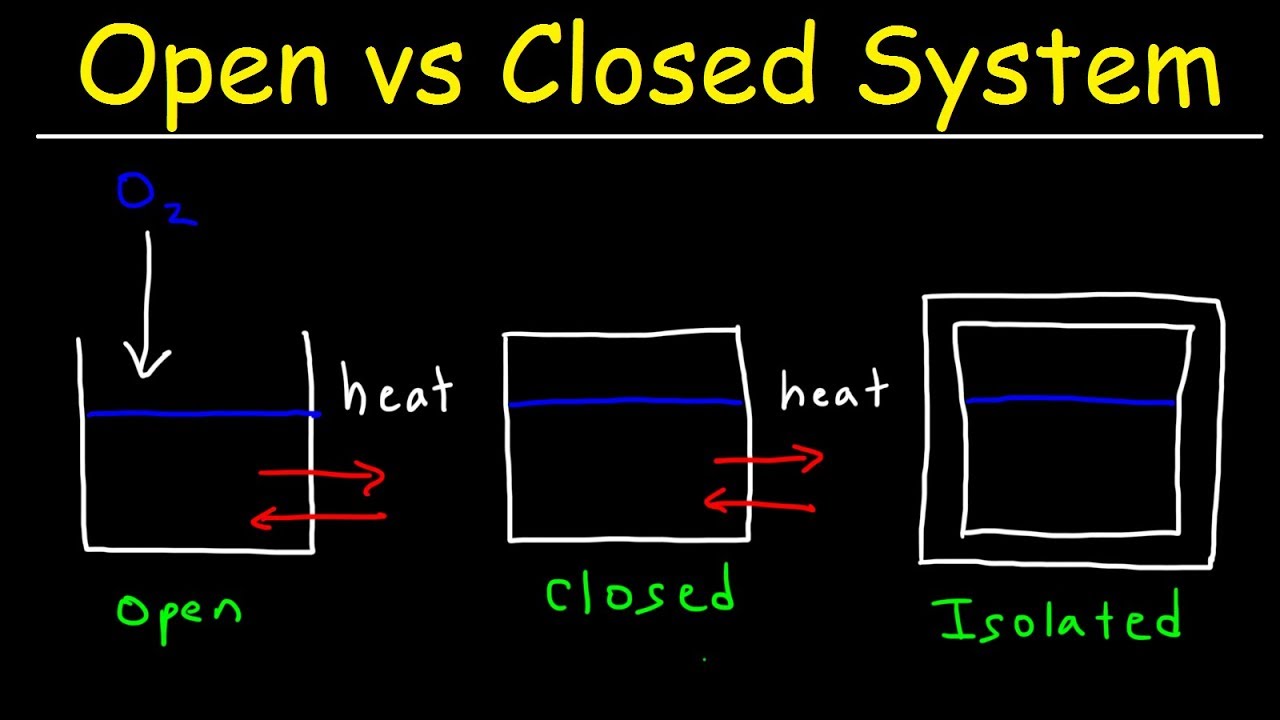
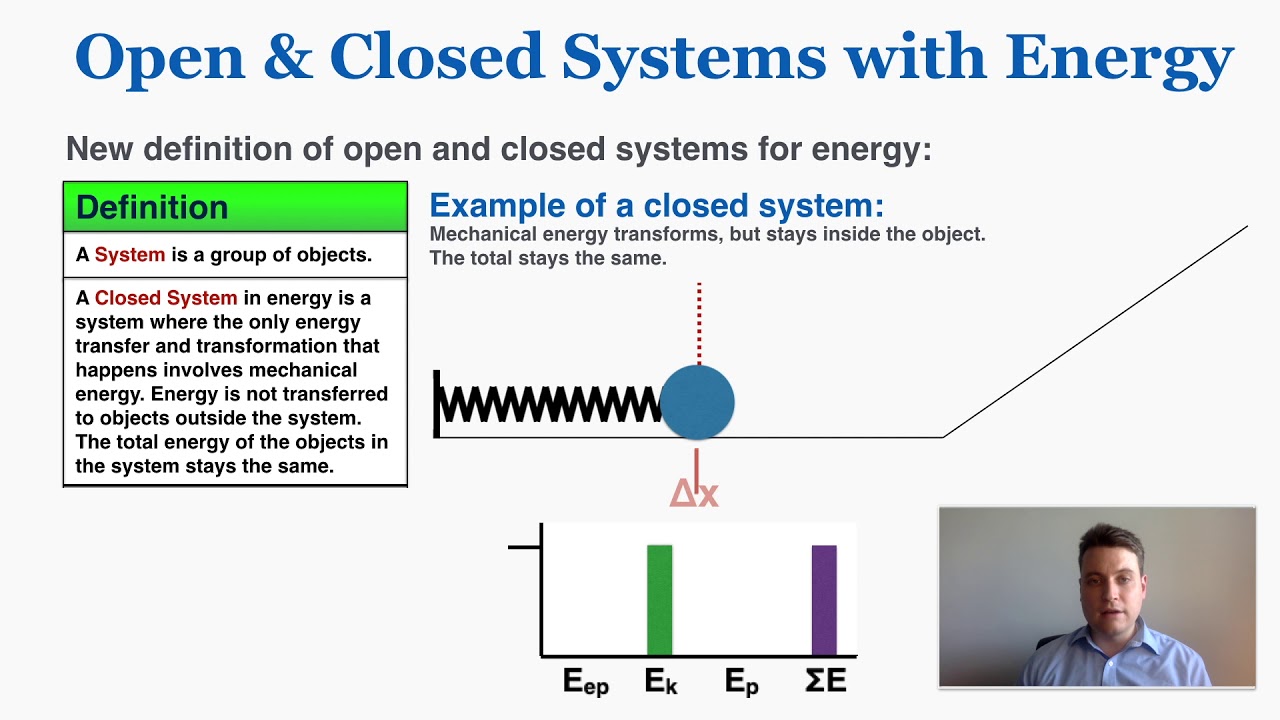
Define and explain the following : (1) Open system (2) Closed system (3) Isolated system. - Sarthaks eConnect | Largest Online Education Community

Define a system and simplify the open-system energy balance for each of the following cases - YouTube
![SOLVED: Thermodynamics Q 1. [6 points]. Define open, closed and isolated systems, respectively. 2 [4 points]. Define equilibrium and steady state in the context of thermodynamics. 3. [8 points]. Define intrinsic and SOLVED: Thermodynamics Q 1. [6 points]. Define open, closed and isolated systems, respectively. 2 [4 points]. Define equilibrium and steady state in the context of thermodynamics. 3. [8 points]. Define intrinsic and](https://cdn.numerade.com/ask_images/59312283ca694f71875b1678d30029d7.jpg)
SOLVED: Thermodynamics Q 1. [6 points]. Define open, closed and isolated systems, respectively. 2 [4 points]. Define equilibrium and steady state in the context of thermodynamics. 3. [8 points]. Define intrinsic and

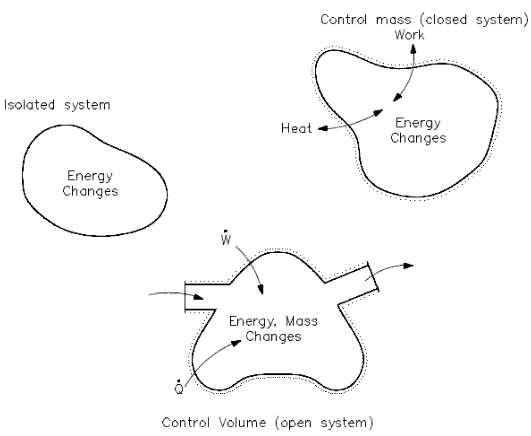
Lesson 4 THERMODYNAMIC SYSTEMS AND PROCESSES DESCRIBE the following types of thermodynamic systems: – Isolated system – Closed system – Open system DEFINE. - ppt download